Return on Investment for 505(b)(2) Products: Is an “AB” Rating Possible?
As new generic product options become more scarce and the competition more fierce, generic manufacturers have recently become very interested in the 505(b)(2) regulatory pathway. The benefits of the 505(b)(2) pathway are well known, such as a potentially lower cost and accelerated path to market. But there are also ways to reduce generic competition for 505(b)(2) products.
One of the common misconceptions is that a drug approved via the 505(b)(2) pathway cannot receive an “AB” therapeutic equivalence (TE) code from FDA. Luckily, this is not true. As such, the sponsor of a product with an “AB” code can expect less generic competition, and greater delays in competing generic approvals. This allows for a greater and longer return on investment. So what is FDA’s TE Code rating system and how many 505(b)(2)’s have been rated “AB”?
TE codes are published in the Orange Book. They were developed to inform Sponsors if FDA has evaluated an approved product as therapeutically equivalent to other pharmaceutically equivalent products, and if so under what circumstances. FDA’s TE codes are used by pharmacists and third party payers including Medicare to determine if a product may be substituted for an innovator product. Products for which the first letter of the TE code is “A” may be substituted, and products with a “B” may not.
Among the products which may be substituted, there is further distinction. If the second letter is a “B”, for example an “AB” rating, a study will be required to compare any proposed generic competitor with the approved product (see Table 1). This may be either in vitro or a clinical pharmacokinetic study comparing the proposed and approved products. Given the time and cost involved in conducting such studies, and the inability of a generic manufacturer to obtain the approved product for the study until it is marketed and available, generic competition for an “AB” rated product is often non-existent for the first few years.
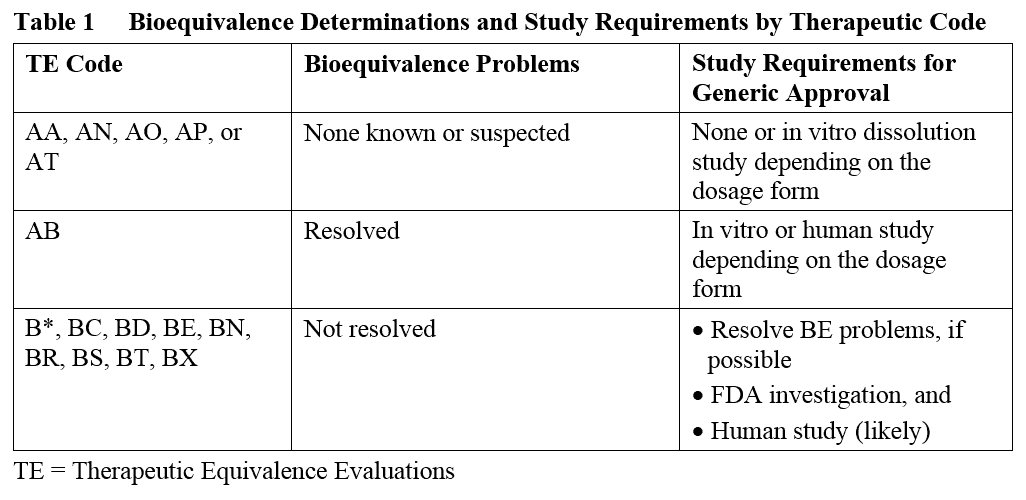
So what percentage of products approved via the 505(b)(2) pathway have received AB codes to date? Of the 83 products for which a TE code has been assigned, a similar number of 505(b)(2) products received an AB code (n = 35) as the combined number of products receiving AA, AN, AO, AP, or AT codes (n = 36). The remaining products received BC (n = 2) or BX (n = 10) codes (see Figure 1).
An example of a product approved via the 505(b)(2) pathway that was assigned an AB code is Metrogel (metronidazole; 1% strength; NDA 21,789; Dow Pharmaceutical Sciences). Metrogel, indicated for the treatment of rosacea, was the first metronidazole gel product in a non-alcohol-based dosage form. The first and only generic for Metrogel (1% strength) was approved 6 years later.
So which products are more likely to get an AB rating? Products approved via the 505(b)(2) pathway that received AB ratings include solid oral dosage forms (n = 26), including 7 extended release products, topical products (n = 8) and 1 oral suspension. These products were approved via the 505(b)(2) pathway because of changes to the dosage form, indication, excipients, strengths, and also new combinations of previously approved products. Products that received AA, AN, AO, AP, or AT codes were typically oral solutions, injectables, and ophthalmics.
While it may be becoming more difficult to get FDA to assign a TE code for 505(b)(2) products, Premier Consulting has the right answers on how to improve the chances that your product will be assigned an AB code, thereby reducing generic competition.
Author:
Angela Drew, PhD
Contributor: