Pre-IND Meetings: The Increasing Frequency of Written Responses Only, and Which FDA Divisions Use Them the Most
The provision of Written Responses Only (WRO) by the FDA in response to Pre-IND meeting requests is here to stay. While rarely requested or preferred by Sponsors, this meeting response format is being used with increasing frequency by many Divisions at the FDA since the last reauthorization of the Prescription Drug User Fee Act (PDUFA V) was signed into law. More than ever, getting a face-to-face Pre-IND meeting requires the preparation of a compelling and complete meeting request and submission package.
WRO address the FDA’s problem of trying to schedule large numbers of staff within the timeframes of the PDUFA performance goals:
- Pre-IND meeting scheduling within 21 days, and
- formal meeting within 60 days of meeting request receipt.
The FDA’s Center for Drug Evaluation and Research (CDER) received almost 3000 requests for formal meetings in 2016, a number that has been increasing annually since 2011. Approximately 95% of those requests are honored through face-to-face meetings, preliminary responses, or WRO. The remainder are denied for various reasons. The difficulty in scheduling 10 – 20 CDER employees, often from different Offices and Divisions (and locations), for each face-to-face meeting is clear.
However, Sponsors who are allocated WRO to their Pre-IND meeting requests can lose a valuable opportunity to discuss the FDA’s responses and obtain clarification on the FDA’s thinking. Thorough preparation for a Pre-IND meeting, including consideration of contingencies, can further refine the feedback obtained from the FDA.
When Did WRO Begin?
PDUFA was created by Congress in 1992, and allows FDA to collect user fees from companies requiring FDA review services for their human drug and biological products. In the 2013 – 2017 Reauthorization of PDUFA (commonly referred to as PDUFA V) that was signed into law in 2012, the Reauthorization Performance Goals And Procedures Fiscal Years 2013 Through 2017 provides examples of Type B and Type C meeting formats of face-to-face, teleconference, videoconference, or written response.
Prior to this, the PDUFA Reauthorization Performance Goals And Procedures Fiscal Years 2008 Through 2012 (PDUFA IV) mentions only ‘face-to-face, teleconference, or videoconference’ options for Type B and Type C meetings.
PDUFA V mentions that a Sponsor may request WRO, rather than a live meeting, or that FDA may determine that WRO may be more appropriate than other meeting formats. This option was also described in the 2015 Draft Guidance for Industry: Formal Meetings Between the FDA and Sponsors or Applicants of PDUFA Products.
However, as far back as 2010, Divisions such as Metabolism and Endocrinology Products; and Anesthesia, Analgesia and Addiction/Rheumatology Products were occasionally providing WRO in response to Pre-IND meeting requests.
WRO are here to stay. In the Commitment letter for PDUFA VI (covering fiscal years 2018 – 2022), a Sponsor request for WRO has been expanded to include any type of meeting (PDUFA V includes this option for Pre-IND and Type C meeting requests). The provision of WRO by the FDA despite a Sponsor’s request for a live meeting remains a listed option for Pre-IND and Type C meetings.
Frequency of Written Responses Only
According to our internal metadata, face-to-face Pre-IND meetings have always been more prevalent than teleconferences or WRO. This continues to be the case since PDUFA V was implemented (2013). However, since 2013, the proportion of 505(b)(2) Pre-IND meetings that received WRO in place of face-to-face or teleconference meetings has remained almost consistently at around 40% (see graph below for our own data). Note that the products include a diverse array of dosage forms and are intended for an extensive range of indications and therapeutic areas.
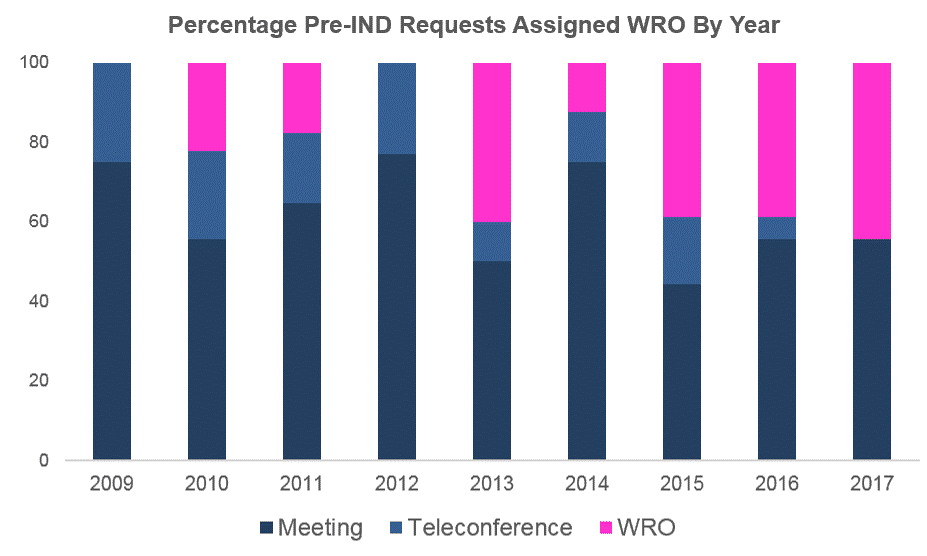 Source: Premier Consulting’s Internal Pre-IND Metadata
Source: Premier Consulting’s Internal Pre-IND Metadata
Note: This data refers only to Pre-IND requests for face-to-face meetings for 505(b)(2) products submitted by Premier Consulting on behalf of Sponsors being represented by Premier Consulting.
Abbreviations: WRO = Written Responses Only
Written Responses Only by Division
Given the large number of Pre-IND meetings that Premier Consulting conducts each year, we have noticed that some Divisions are more likely to use the WRO option than others. In the graph below, we have shown the percent of 505(b)(2) Pre-IND meeting requests submitted by Premier Consulting that received WRO from 2013 (implementation of PDUFA V) until the present. Once again, the products include diverse dosage forms for a broad range of indications and therapeutic areas. The Divisions of Metabolism and Endocrinology Products; Pulmonary, Allergy and Rheumatology; and Neurology Products are the most likely to send WRO in lieu of a face to face meeting. The least likely to assign WRO are the Divisions of Transplant and Ophthalmology Products (all Premier Consulting requests were assigned face-to-face meetings), Oncology Products 1 and 2 (data combined), and Cardiovascular and Renal Products.
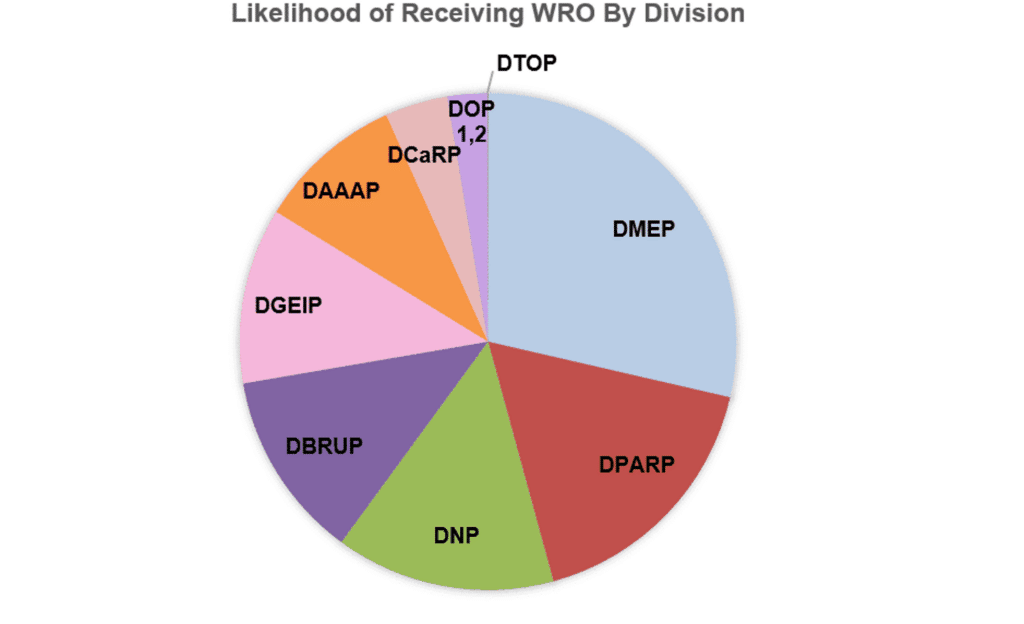 Source: Premier Consulting’s Internal Pre-IND Metadata
Source: Premier Consulting’s Internal Pre-IND Metadata
Note: This data refers only to 505(b)(2) Pre-IND requests for face-to-face meetings submitted on behalf of Sponsors being represented by Premier Consulting. Data are calculated as % of meetings receiving WRO compared with live meetings (face to face and teleconferences) as a proportion of all Pre-INDs prepared by Premier Consulting between 2013 and the present.
Abbreviations: DMEP = Division of Metabolism and Endocrinology Products; DPARP = Division of Pulmonary, Allergy, and Rheumatology Products; NP = Division of Neurology Products; DBRUP = Division of Bone, Reproductive and Urologic Products; DGIEP = Division of Gastroenterology and Inborn Errors Products; DAAAP = Division of Anesthesia, Analgesia, and Addiction Products; CaRP = Division of Cardiovascular and Renal Products; DOP 1,2 = Division of Oncology Products 1,2 (combined); DTOP = Division of Transplant and Ophthalmology Products; WRO = Written Responses Only
Tips for Optimizing Feedback From Written Responses Only
As we frequently blog, there is no substitute for thorough preparation for a Pre-IND Meeting. This includes asking the right questions, providing the right background information in a logical, concise manner, and most importantly for a product intended for the 505(b)(2) pathway, mapping out the development program for FDA feedback. As with any meeting, but especially considering the likelihood of getting WRO, you may wish to include more options for your development plan .
If the FDA doesn’t agree with your proposed development plan, more options will allow for more feedback. This will allow you to move forward with your IND-opening study as soon as possible. Consider proposing alternatives that will best fulfill your program goals and satisfy the FDAs requirements. And map out for the FDA how your proposal meets current standards.
Remember: it is your meeting, so be sure to get as much information as you need to confirm and initiate your development program. Your fastest path to market will be obtaining sufficient clarity so you don’t need to request a new meeting, and in gaining the knowledge that FDA agrees with your general development plan.
If you would like to discuss how to get the best outcome from your 505(b)(2) Pre-IND meeting, or to get advice on your drug/device development program, contact us.
Author:
Angela Drew, PhD
Product Ideation Consultant