Faster Approval of Combination Drug Products Via the 505(b)(2) Pathway
On April 11 and 12, 2016, Ken Phelps, Strategic Advisor at Premier Consulting, spoke at the 505(b)(2) Forum and CTrials Conference in Tel Aviv, Israel, on the topic of 505(b)(2) Combination Drug Products. What is attractive about the 505(b)(2) regulatory pathway, and specifically, why is it so beneficial for approval of combination products? We’d like to open up and share a part of Ken’s recent discussion here.
What Is Special About the 505(b)(2) Approval Pathway?
Under United States law, “a 505(b)(2) application is one for which one or more of the investigations relied upon by the applicant for approval ‘were not conducted by or for the applicant and for which the applicant has not obtained a right of reference or use from the person by or for whom the investigations were conducted’” (21 U.S.C. 355(b)(2)). This means the 505(b)(2) regulatory pathway can be used to leverage information that already exists in order to reduce the number and size of studies required for approval.
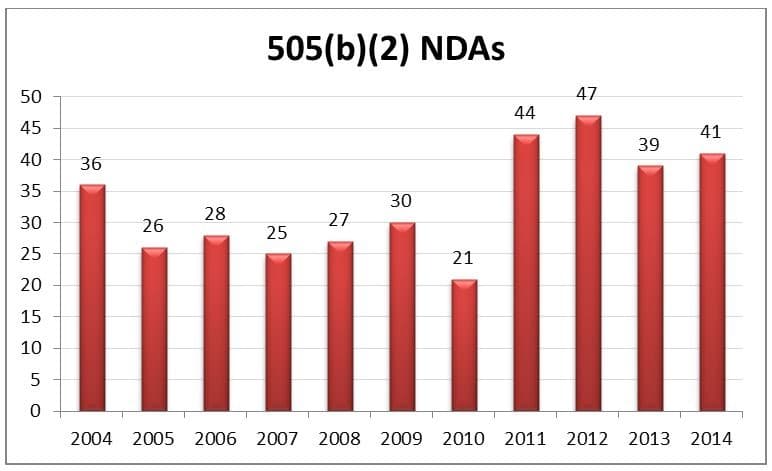
The graphic above shows that over the past ten years, the rate of 505(b)(2) submissions have been increasing such that between the period of 2004 – 2014, more than 350 new drugs have been approved by the FDA through the 505(b)(2) pathway. As they utilize existing information on a drug, 505(b)(2) products typically have a lower risk, lower cost, and faster time to market than traditional new drug approvals.
Examples of combination products for which the 505(b)(2) pathway may be appropriate include new combinations, new dosage forms of approved combinations, and new molecular entities (one or both drugs has not been approved). Two examples of products approved via the 505(b)(2) pathway include:
- Aripiprazole lauroxil (new molecular entity and drug-device combination)
- RLD = aripiprazole (Abilify®, Otsuka NDA 021436), oral tablets/solution for daily dosing
- New molecular entity: aripiprazole lauroxil (Aristada™, Alkermes Inc.; prodrug of the prodrug of aripiprazole), extended-release suspension in prefilled syringes for monthly or 6-weekly dosing, approved 2015.
- Lidocaine and tetracaine (new combination)
- Previous approvals: Lidocaine- approved prior to 1982; Tetracaine – not approved
- New combination: Lidocaine + Tetracaine (Synera, Galen Specialty, NDA 021623) approved 2005.
Which Types of Combination Drug Products are Eligible for Approval via the 505(b)(2) Pathway?
- A drug plus a device
- Two drugs and a device
- A drug plus a biologic
- A drug-drug combination
In each of the first 3 cases, the products are only appropriate for the 505(b)(2) pathway if the FDA agrees that the Primary Mode of Action (PMOA) of the entire product is the drug.
Examples of 505(b)(2)-appropriate combination drug products include:
- Monoclonal antibody combined with a therapeutic drug (if drug is PMOA)
- Pre-filled syringes
- Insulin injector pens
- Metered-dose inhalers
- Transdermal patches
- Drug packaged with a delivery device
- Two drugs combined in a fixed-dosage form
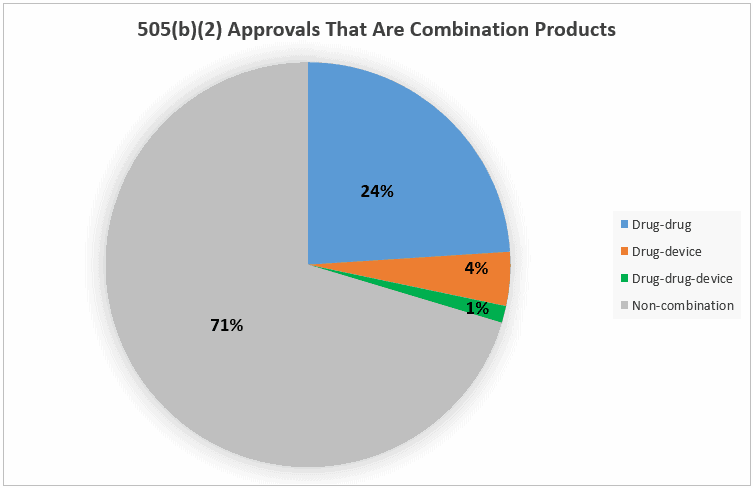
The figure above shows the 505(b)(2) approvals by type. Note the blue slice, 24%, which represents products registered as new drug-drug combination products. When we include drug-device combinations and products for which one component was a new molecular entity, the figure rises to 29.7% of all approved 505(b)(2) products that are some type of combination product.
Here at Premier Consulting, we believe combination products are a growing area filled with potential. Contact us to learn more about 505(b)(2) combination products, and find out more about how Premier Consulting can help you and your business reach your drug development goals.
Author:
Angela Drew, PhD
Product Ideation Consultant